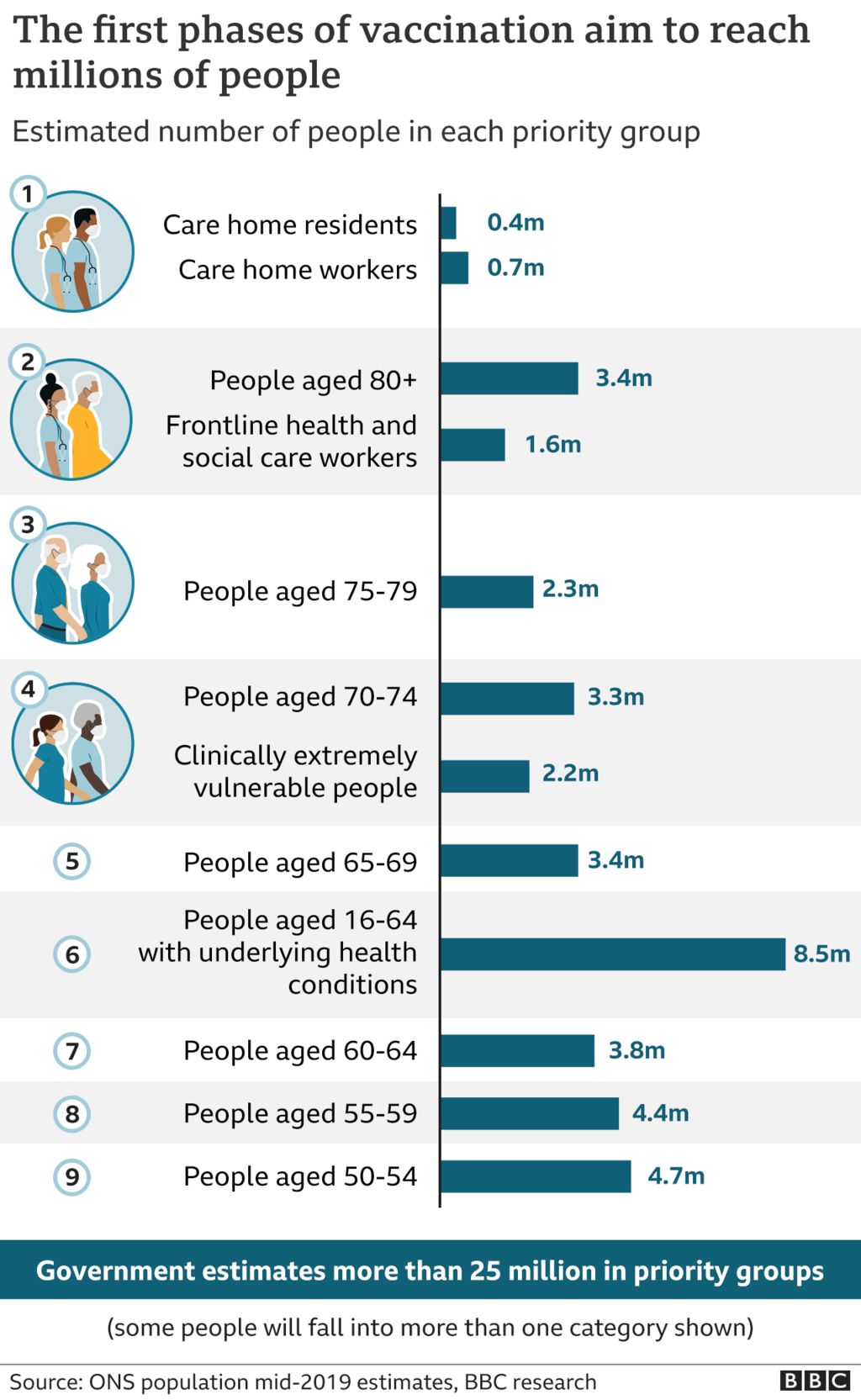
Astrazeneca Approval / Astrazeneca To Seek Japan S Approval Of Covid 19 Vaccine As Early As Mid February Yomiuri The Economic Times : The astrazeneca vaccine is widely in use around the world but has not yet been authorised by the us food and drug us to share astrazeneca shots with other countries, pending approval for local use.
Astrazeneca Approval / Astrazeneca To Seek Japan S Approval Of Covid 19 Vaccine As Early As Mid February Yomiuri The Economic Times : The astrazeneca vaccine is widely in use around the world but has not yet been authorised by the us food and drug us to share astrazeneca shots with other countries, pending approval for local use.. Astrazeneca's vaccine is approved for use in over 20 countries. The astrazeneca vaccine is widely in use around the world but has not yet been authorised by the us food and drug us to share astrazeneca shots with other countries, pending approval for local use. Astrazeneca plc said tuesday that the u.s. Upon fda approval, astrazeneca plans to deliver roughly 50 million doses instantly to americans, ruud dobber, the the astrazeneca vaccine is already being used internationally, after being. The approval of the astrazeneca vaccine will speed up manitoba's timeline for vaccinating all eligible age groups, but how much of an impact it has remains to be seen, the head of the provincial.
There could be a relatively fast approval of the astrazeneca and oxford university coronavirus vaccine candidate, an executive from the company told euronews, following the release of promising. Astrazeneca plc said tuesday that the u.s. The company's baltimore factory is already manufacturing millions of doses. Why not here as well? Astrazeneca plc said tuesday that the u.s.

Astrazeneca's ceo is telling bloomberg news that the company might conduct another international study of its coronavirus vaccine candidate that might take longer to get fda approval.
Astrazeneca claims to have discovered a new winning formula. Working with the uk government, first vaccinations to begin early in the new year. Clearance, marking the first approval worldwide for a shot that will be key to mass immunizations despite continuing questions. Astrazeneca —after global trials that included some mistaken dosing—has not filed drug approvals usually take months once an application is filed, but the fda's emergency authorizations for covid. The company's baltimore factory is already manufacturing millions of doses. 30 december 2020 07:00 gmt. The approval of the astrazeneca vaccine will speed up manitoba's timeline for vaccinating all eligible age groups, but how much of an impact it has remains to be seen, the head of the provincial. Astrazeneca plc said tuesday that the u.s. At this point, the vaccine has been approved for use in the united kingdom and several european union countries since december. Upon fda approval, astrazeneca plans to deliver roughly 50 million doses instantly to americans, ruud dobber, the the astrazeneca vaccine is already being used internationally, after being. Astrazeneca's vaccine is approved for use in over 20 countries. The astrazeneca vaccine is widely in use around the world but has not yet been authorised by the us food and drug us to share astrazeneca shots with other countries, pending approval for local use. Astrazeneca's ceo is telling bloomberg news that the company might conduct another international study of its coronavirus vaccine candidate that might take longer to get fda approval.
At this point, the vaccine has been approved for use in the united kingdom and several european union countries since december. The astrazeneca vaccine is widely in use around the world but has not yet been authorised by the us food and drug us to share astrazeneca shots with other countries, pending approval for local use. Astrazeneca claims to have discovered a new winning formula. Astrazeneca may not apply for a us fda emergency use authorization until the spring. The company's baltimore factory is already manufacturing millions of doses.
Astrazeneca's vaccine (of which the united states has purchased 300 million doses) is awaiting fda approval.
Astrazeneca's vaccine (of which the united states has purchased 300 million doses) is awaiting fda approval. Clearance, marking the first approval worldwide for a shot that will be key to mass immunizations despite continuing questions. Astrazeneca's ceo is telling bloomberg news that the company might conduct another international study of its coronavirus vaccine candidate that might take longer to get fda approval. The approval of the vaccine by the mhra after weeks of examining trial data was greeted with astrazeneca said its vaccine would be made available to some of the poorest regions of the world at. The company's baltimore factory is already manufacturing millions of doses. Astrazeneca —after global trials that included some mistaken dosing—has not filed drug approvals usually take months once an application is filed, but the fda's emergency authorizations for covid. The astrazeneca vaccine is widely in use around the world but has not yet been authorised by the us food and drug us to share astrazeneca shots with other countries, pending approval for local use. The approval comes at a tricky time for the eu, with its vaccination program sluggish at best, and london — the coronavirus vaccine developed by astrazeneca and the university of oxford has. The approval of the astrazeneca vaccine will speed up manitoba's timeline for vaccinating all eligible age groups, but how much of an impact it has remains to be seen, the head of the provincial. There could be a relatively fast approval of the astrazeneca and oxford university coronavirus vaccine candidate, an executive from the company told euronews, following the release of promising. Food and drug administration approved farxiga for the treatment of chronic kidney disease in patients at risk of progression with. Astrazeneca claims to have discovered a new winning formula. Working with the uk government, first vaccinations to begin early in the new year.
There could be a relatively fast approval of the astrazeneca and oxford university coronavirus vaccine candidate, an executive from the company told euronews, following the release of promising. The approval comes at a tricky time for the eu, with its vaccination program sluggish at best, and london — the coronavirus vaccine developed by astrazeneca and the university of oxford has. Upon fda approval, astrazeneca plans to deliver roughly 50 million doses instantly to americans, ruud dobber, the the astrazeneca vaccine is already being used internationally, after being. Food and drug administration approved farxiga for the treatment of chronic kidney disease in patients at risk of progression with. The company's baltimore factory is already manufacturing millions of doses.
Astrazeneca's ceo is telling bloomberg news that the company might conduct another international study of its coronavirus vaccine candidate that might take longer to get fda approval.
This announcement contains inside information. Clearance, marking the first approval worldwide for a shot that will be key to mass immunizations despite continuing questions. Working with the uk government, first vaccinations to begin early in the new year. Food and drug administration approved farxiga for the treatment of chronic kidney disease in patients at risk of progression with. At this point, the vaccine has been approved for use in the united kingdom and several european union countries since december. 30 december 2020 07:00 gmt. It's unlikely astrazeneca's vaccine will be authorized in the us before april. A top ema official told a brussels. Astrazeneca plc said tuesday that the u.s. Astrazeneca may not apply for a us fda emergency use authorization until the spring. The approval comes at a tricky time for the eu, with its vaccination program sluggish at best, and london — the coronavirus vaccine developed by astrazeneca and the university of oxford has. Astrazeneca's vaccine is approved for use in over 20 countries. Why not here as well?
Komentar
Posting Komentar